The drug companies Pfizer and BioNTech* say the coronavirus vaccine they’re developing seems to be 90% successful at preventing people from getting Covid-19. The results aren’t final, but the news is much better than many experts expected.
Coronavirus Vaccines
COVID-19 is caused by a virus called SARS-CoV-2. The virus has changed life around the world, making over 51 million people sick, and killing more than 1.25 million people.
Normally, vaccines take years to develop, but because the coronavirus is so serious, people everywhere are trying to speed up the process.
Currently, over 320 different vaccines are being studied. Before vaccines are used widely, they normally go through several steps of testing, called phases. Phase 3 is the last, and most important phase.
Right now, 10 vaccines are in Phase 3 trials. Once a vaccine has passed all of the needed tests, it can be approved for use.

(Source: AnToonz, via Flickr.com.)
The 90% success rate may change, because the vaccine’s trial isn’t over. Still, the high numbers are encouraging. The US government planned to approve any vaccine with more than 50% success. This is the first vaccine to pass that goal.
“Today is a great day for science and humanity,” said Dr. Albert Bourla, Pfizer’s leader.
Over 43,000 people took part in this Phase 3 trial. Around half were given the vaccine, which comes as two shots, given three weeks apart. The other half got shots that didn’t contain the vaccine.
😕
This image has not been loaded because of your cookie choices. To view the content, you can accept 'Non-necessary' cookies.
Over 43,000 people took part in this Phase 3 trial. Around half were given the vaccine, which comes as two shots, given three weeks apart. The other half got shots that didn’t contain the vaccine.
To make sure the results were accurate, neither the people being tested or their doctors were told whether they got the vaccine or not. The company says the vaccine hasn’t caused serious problems for anyone who got it.
So far, 94 of the people in the trial have gotten the coronavirus. Almost all the cases were in people who didn’t get the vaccine.
Pfizer has to wait for two months after everyone gets both shots before asking the US government for special early approval for the vaccine. The company plans to ask for early approval in the third week of November.
😕
This image has not been loaded because of your cookie choices. To view the content, you can accept 'Non-necessary' cookies.
Pfizer has to wait for two months after everyone gets both shots before asking the US government for special early approval for the vaccine. The company plans to ask for early approval in the third week of November.
If the government approves the vaccine quickly, it’s possible that some people – probably health care workers – could get the vaccine by the end of the year.
Even if the vaccine gets early approval, the trial won’t end until 164 people have gotten Covid-19. The current results are just an early report. There aren’t many details, and the trial hasn’t been reviewed by outside health experts. That will come once the trial is over.
Several questions remain about the Pfizer/BioNTech vaccine, such as whether it protects against all forms of the coronavirus, whether it protects people who have already had the coronavirus, and how long its protection will last.
(Source: Kojach, via Flickr.com.)
It will also take time to learn whether the vaccine causes problems that may show up later.
The Pfizer/BioNTech vaccine is an mRNA vaccine – a new kind of vaccine that hasn’t been approved for general use before.
These vaccines rely on packaging up part of the coronavirus RNA – the special code that helps create the coronavirus. In this case, the mRNA vaccines help the body recognize and fight the spikes on the outside of the coronavirus.
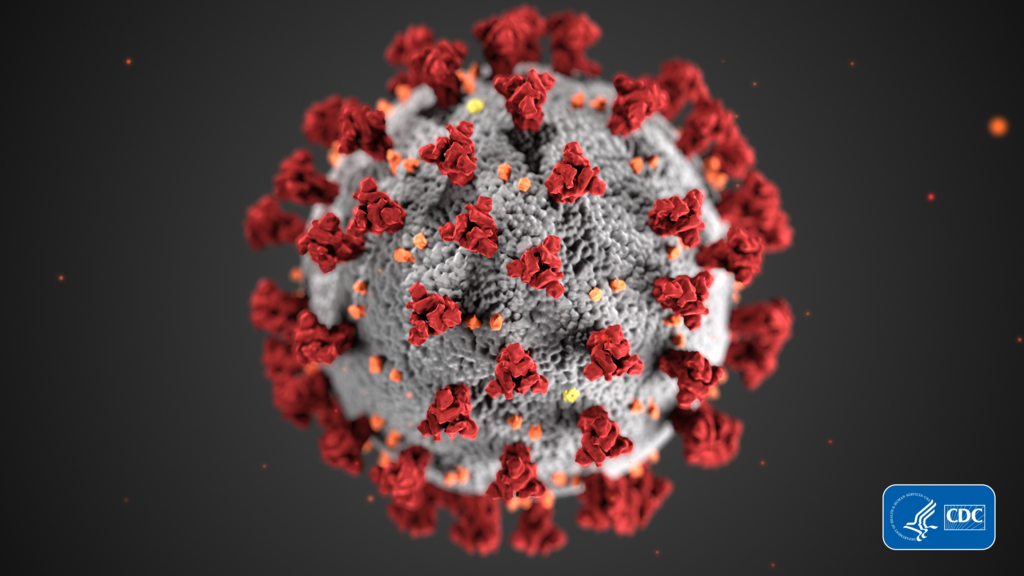
(Source: CDC/ Alissa Eckert, MS; Dan Higgins, MAMS [Public domain], via Wikimedia Commons.)
Even though the vaccine isn’t approved yet, the companies are already producing it, to save time if it is approved. They expect to make enough to protect 25 million people this year, and enough for 650 million people next year.
Countries around the world have ordered millions of doses of the vaccine. But safely delivering the vaccine will be a challenge. That’s because it needs to be kept extremely cold: -94º Fahrenheit (-70º Celsius).
😕
This image has not been loaded because of your cookie choices. To view the content, you can accept 'Non-necessary' cookies.
Safely delivering the vaccine will be a challenge because it needs to be kept extremely cold. The transport company UPS is building huge freezers to help keep vaccines cold as they’re being delivered. This will be hard in places without reliable electricity.
Pfizer has developed special coolers to move the vaccine. Even so, it will be hard to store and move the vaccine in places without reliable electricity.
* Pfizer is an American company. BioNTech is a German company.
